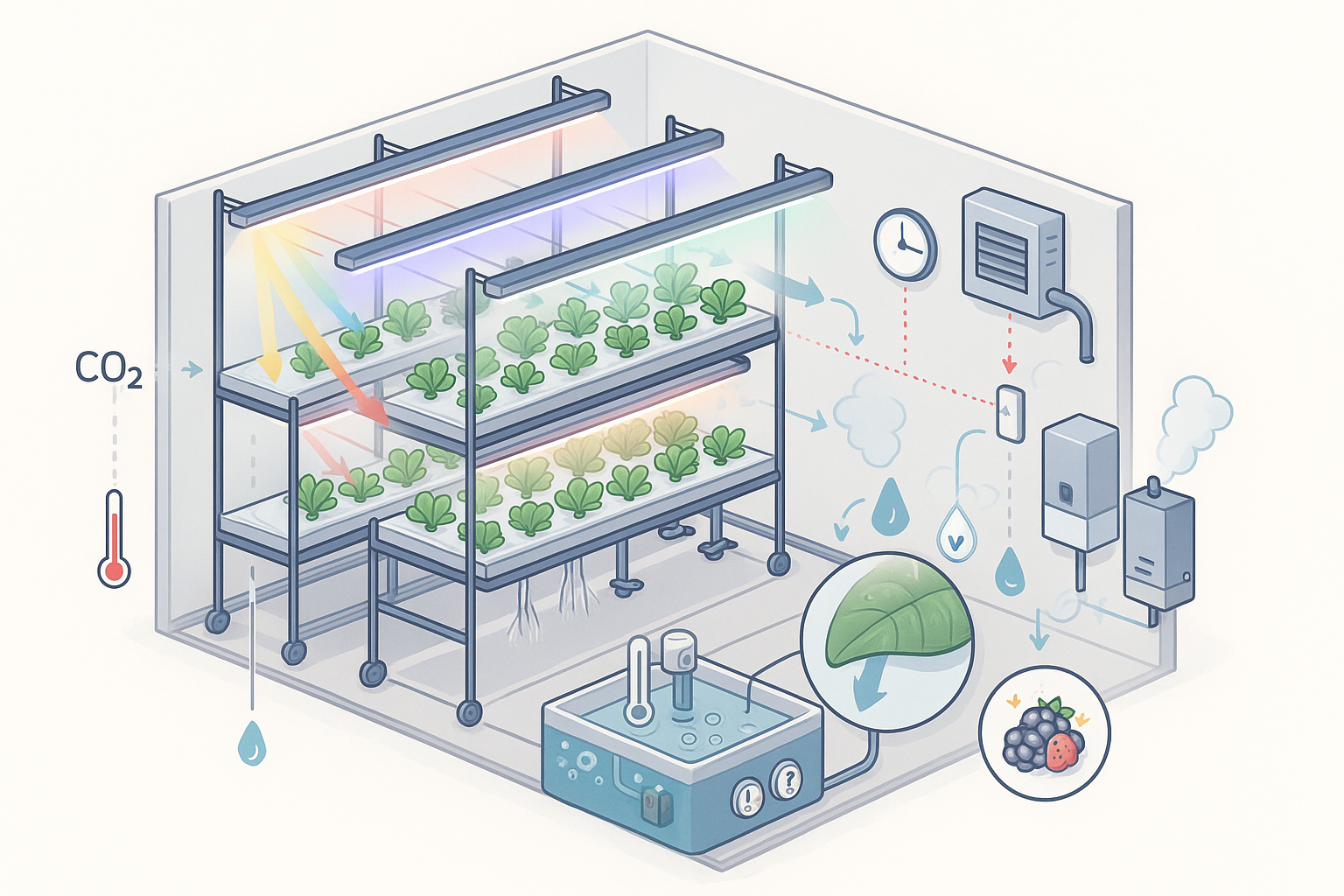
Introduction: why physiology matters in CEA
Plant physiology in CEA is not an abstract academic subject: it is the operating manual for yield, quality and cost. In a controlled environment, light, carbon dioxide, temperature, humidity, water chemistry and nutrients are all set by design choices. Each decision pushes plant metabolism in a direction: faster photosynthesis or higher respiration; compact architecture or shade-avoiding stretch; sweeter leaves or more biomass. A grasp of the underlying processes allows growers, researchers and investors to design systems that deliver predictable outcomes rather than relying on rules of thumb.
Light: photons, spectrum and dose
Photosynthesis is driven by the number of photons plants intercept per day. The two practical levers are instantaneous intensity, usually expressed as PPFD in umol m-2 s-1, and the accumulated daily light integral (DLI) in mol m-2 d-1. For leafy greens, a modest PPFD sustained over longer photoperiods can achieve the same DLI as brief high intensity. DLI is therefore the anchor metric for planning tiers, fixture spacing and runtime. University extension sources treat DLI as a primary control variable for growth rate and quality, and give workable ranges for many crops.
Spectral quality matters because pigments and photoreceptors partition tasks. Blue photons guide stomata and morphology, red photons are highly efficient for photosynthesis, and green photons penetrate thick canopies. Far-red photons between 700 and 750 nm deserve special attention in indoor farms: alone they do little, yet when added to 400 to 700 nm light they increase canopy quantum yield and leaf expansion, improving radiation capture and often yield. This evidence has prompted calls to include far-red within the operational definition of photosynthetic photons for crop lighting.
Carbon dioxide: the second substrate of photosynthesis
At a given light dose, raising CO2 concentration usually increases net photosynthesis in C3 crops until other factors become limiting. Meta-analyses of free-air CO2 enrichment experiments report average yield gains of roughly one fifth for C3 species at about 200 ppm above ambient, with variation by crop and nutrient status. These are field and greenhouse figures; the controlled, high-light conditions of plant factories often reveal larger marginal returns up to a point, after which CO2 becomes wasteful or creates safety concerns. The operational lesson is simple: pair CO2 set-points with the actual light dose and canopy development, rather than fixing a single value for all stages.
Temperature and respiration: rate controls and costs
Temperature accelerates both photosynthesis and respiration, but the balance between them decides whether extra heat buys growth or merely burns carbon. A common shorthand says respiration doubles for each 10 oC rise. In reality the temperature coefficient Q10 is not constant, and typically declines as leaves warm. Acclimation further shifts these responses over days. For design and scheduling, thermal time or growing degree days provides a practical clock for development, provided confounding factors such as photoperiod and nutrient stress are considered. In practice, many indoor farms benefit from slightly cooler night temperatures to restrain respiratory losses while maintaining daytime warmth to support photosynthesis.
Humidity, vapour pressure deficit and the leaf energy balance
Water relations link climate control to gas exchange. Vapour pressure deficit, VPD, reflects the drying power of air: high VPD pulls water rapidly from leaves, low VPD slows transpiration and can impede calcium delivery and cooling. Both extremes reduce performance. Leaf energy balance theory shows that transpiration is the chief passive cooling pathway: if VPD or stomatal conductance limit it, leaf temperature rises above air temperature and photosynthesis suffers. Experimental work in protected systems indicates that many crops perform well when VPD is held near the lower middle of the commonly cited range for growth, and that limiting rapid VPD swings helps to maintain open stomata and stable assimilation. The practical advice is to set coordinated temperature and humidity targets that keep VPD in a moderate band and avoid abrupt transitions during photoperiod changes.
Roots, oxygen and nutrients: the hidden half of CEA
Hydroponic roots occupy a managed fluid environment. Dissolved oxygen supports respiration and ion uptake at the root surface, and inadequate oxygen supplies can restrict growth, especially at higher solution temperatures. Classic work in lettuce showed surprisingly low critical thresholds under the tested conditions, yet subsequent studies across species and systems confirm that oxygenation remains a yield lever in recirculating tanks and channels. The safest approach is to prevent warm, stagnant solution layers and maintain vigorous turnover and gas exchange rather than chasing a single ppm target.
Nutrient concentration and pH define availability and ionic balance. Electrical conductivity is a convenient proxy for total salts; crop-specific EC set-points should be modulated by stage, light and temperature because high light and warm conditions increase demand. Extension guidance for lettuce suggests EC around 1.4 to 1.8 mS cm-1 with pH near 6.0 to 7.0, which aligns with many commercial recipes in plant factories. The form of nitrogen also matters: modest inclusion of ammonium alongside nitrate can enhance growth and reduce nitrate accumulation in leaves, although high ammonium fractions increase the risk of toxicity under suboptimal pH or low oxygen (see Sandoya et al., 2021).
Photoperiod and the plant clock: timing as a control variable
The same photons that power photosynthesis also set the calendar. Photoperiod interacts with the circadian system to regulate flowering, tuberisation and architecture through pathways such as CONSTANS and FT (Shim et al., 2017). For photoperiod-sensitive species, night-interruption or extended-day lighting at very low intensities can shift development without materially changing DLI. This separation of energy and timing is often underused in CEA design. It allows growers to schedule flowering and vegetative growth independently of photosynthetic light budgets, saving power while keeping crops on target.
Quality and secondary metabolism: beyond biomass
Nutritional and sensory quality reflects both genetics and environment. Light dose and spectrum can alter pigments, aromatic compounds and nitrate levels; temperature and VPD steer calcium transport and texture; nitrogen form and EC shape flavour. Physiological control therefore extends beyond grams per square metre: it is the means by which indoor farms tailor produce to a market brief, for example higher anthocyanins or consistent crunch at a defined harvest age. The research base here is diverse; the common thread is that quality traits respond to the same levers already needed for yield, so well-designed environments deliver both.
Putting the principles to work: a simple integration example
Consider a three-tier leafy-green room built for steady weekly harvests. Start by defining DLI from the required crop time. Choose a PPFD and photoperiod that fit heat loads, worker access and energy tariffs. Set CO2 to match light and canopy closure. Choose temperature bands that maintain a positive carbon balance after accounting for respiration, then coordinate humidity so that VPD sits in a moderate range through lights-on and lights-off transitions. Keep the solution cool enough for oxygen supply or provide agitation and aeration. Hold EC and pH within crop-appropriate ranges, adjusting with stage and season. Use photoperiod control where species are sensitive, for example to keep basil vegetative while pushing biomass. Measure responses and adjust one lever at a time. The physiology tells you what to change next, and why it will work.
Conclusion: physiology as the foundation of reliable CEA
Indoors, the plant’s environment is a product, not a constraint. Plant physiology in CEA provides the structure to design that product with intention: photons and carbon for assimilation, temperature and VPD for balance, roots, oxygen and ions for uptake, and photoperiod for timekeeping. The reward is not only higher yield, but greater predictability and market-ready quality. The science is mature enough to guide day-to-day choices, but the art lies in integrating these controls for the crop in front of you.
Bibliography
Ainsworth E A and Long S P. 2005. What have we learned from 15 years of FACE. New Phytologist 165:351-371. PubMed
Ainsworth E A and Rogers A. 2021. Thirty years of free-air CO2 enrichment. Global Change Biology 27:27-49. PubMed
Atkin O K and Tjoelker M G. 2003. Thermal acclimation and the dynamic response of plant respiration to temperature. Trends in Plant Science 8:343-351. PubMed
Currey C J, Lopez R G and Runkle E S. Managing photoperiod in the greenhouse. Purdue Extension HO-253-W. extension.purdue.edu
Inoue T et al. 2021. Minimising VPD fluctuations maintains higher stomatal conductance and photosynthesis. Frontiers in Plant Science 12:646144. PMC
ISHS. Effect of dissolved oxygen concentration on lettuce growth in floating hydroponics. Evidence on DO thresholds. PubMed
Monteith J L and Unsworth M H. Principles of Environmental Physics. Elsevier. Concepts of leaf energy balance and VPD. denning.atmos.colostate.edu
Purdue Extension. Measuring Daily Light Integral in a Greenhouse. HO-238-W. extension.purdue.edu
Runkle E and Lopez R G. E-Gro and allied factsheets on DLI and lighting control. e-gro.org
Sandoya et al. 2021. Growing Lettuce in Small Hydroponic Systems. Askifas
Shim, J. S., Kubota, A., & Imaizumi, T. (2017). Circadian clock and photoperiodic flowering in Arabidopsis: CONSTANS is a hub for signal integration. Plant physiology, 173(1), 5-15.
UF IFAS. Growing Lettuce in Small Hydroponic Systems. EDIS HS1422. EC and pH guidance. Ask IFAS - Powered by EDIS
Zhen S and Bugbee B. 2020 and 2021. Far-red photons and photosynthesis at the canopy scale. Frontiers in Plant Science; Plant Physiology perspectives. Frontiers+1
Notes on interpretation
References provide the mechanistic basis and quantitative ranges. Translate them to practice through measurement, small controlled adjustments and crop-specific trials; physiology gives the direction of travel, local data give the scale.